COVID-19 News Key Updates
Ministry of Health COVID-19 Information
You can visit the MOH website for the latest Singapore COVID-19 updated situation news.
26 Oct 2020
MOH ADVISES TEMPORARILY CEASING USE OF 2 FLU VACCINES AFTER DEATHS IN SOUTH KOREA
SINGAPORE: The Ministry of Health (MOH) has advised that the use of two influenza vaccines in Singapore be "temporarily ceased" as a precautionary measure after deaths were reported in South Korea following vaccinations.
South Korean health authorities said they have found no direct link between the deaths and the flu shots.
"This is a precautionary measure following reported deaths after influenza vaccination in South Korea," MOH said in a press release on Sunday (Oct 25).
"No deaths associated with influenza vaccination have been reported in Singapore to date."
Dozens of South Koreans died this month after receiving flu shots as part of a state-run vaccination programme. As of Saturday, 48 deaths had been reported.
On Sunday, MOH said that the ministry and the Health Sciences Authority (HSA) were monitoring the situation closely.
"HSA is in touch with the South Korean authorities for further information as they investigate to determine if the deaths are related to influenza vaccinations.
"Based on information released by the South Korean health authorities, seven brands of influenza vaccines were administered to the individuals involved in the deaths reported in South Korea," the ministry said.
Two of the seven brands are available in Singapore, MOH added.
They are SKYCellflu Quadrivalent, manufactured by SK Bioscience and locally distributed by AJ Biologics; and VaxigripTetra, manufactured by Sanofi Pasteur and locally distributed by Sanofi Aventis.
"As a precautionary measure while HSA is assessing the implications of the reported deaths in South Korea, MOH has informed healthcare providers and medical practitioners to temporarily stop the use of these two vaccines," the ministry said.
It added: "Healthcare providers and medical practitioners may continue to use the two other influenza vaccines that have been brought into Singapore for the Northern Hemisphere 2020/21 influenza season."
FLU VACCINE "GENERALLY SAFE AND WELL TOLERATED"
MOH said that people recommended to receive the influenza vaccination may continue to receive their vaccination using other brands of vaccines.
This included people recommended to receive the vaccination under the National Childhood Immunisation Schedule and National Adult Immunisation Schedule.
The ministry added that the vaccination provided protection against seasonal influenza viruses, and was effective in reducing the risk of complications and deaths due to influenza.
"This is especially so for vulnerable groups such as the elderly, the young, pregnant women and those with certain pre-existing medical conditions," it said.
"Influenza vaccination is generally safe and well tolerated."
The ministry said common side effects from influenza vaccination may include soreness and redness at the injection site, fever, headaches, muscle aches, fatigue and nausea.
"These side effects are generally mild and resolve on their own. In rare instances, a person may experience high fever or severe allergic reactions (such as breathing difficulty, wheezing and swelling around the eyes) and immediate medical attention should be sought," MOH said.
It added that vaccines approved for use in Singapore have been evaluated by HSA to ensure that they meet "required international standards of quality, safety and efficacy".
"To ensure that the benefits continue to outweigh the risks, HSA monitors the safety of vaccines through an adverse event monitoring system.
"It draws on the network of local healthcare professionals and international regulatory counterparts to pick up adverse events suspected to be associated with the vaccines," MOH said.
The ministry said it would continue to assess the situation as more information became available and further advise on the use of the affected vaccines.
"It draws on the network of local healthcare professionals and international regulatory counterparts to pick up adverse events suspected to be associated with the vaccines," MOH said.
The ministry said it would continue to assess the situation as more information became available and further advise on the use of the affected vaccines.
23 Oct 2020
SINGAPORE, GERMANY AGREE TO SET UP 'GREEN LANE' FOR ESSENTIAL BUSINESS AND OFFICIAL TRAVEL
SINGAPORE: Residents in Singapore and Germany may soon be able to travel between the two countries for essential business and official purposes, after both sides agreed to establish a reciprocal green lane.
This was announced in a joint press statement on Friday (Oct 23) by the foreign affairs ministries of both countries.
"The reciprocal green lane will enable essential travel for business or official purposes, via direct flights between both countries," said the statement.
Eligible applicants will have to abide by the COVID-19 prevention and public health measures mutually agreed by both countries, it added.
Both countries also said that the green lane could serve as a model for future similar arrangements between Singapore and other European countries. Operational details of the green lane, including the procedural requirements, health protocols, and application process, will be announced at a later date.
21 Oct 2020
CHILDREN AGED BELOW 5 AT LOWEST RISK OF COVID-19 INFECTION FROM ADULTS: KKH STUDY
SINGAPORE: Children under the age of five are at “lowest risk” of contracting COVID-19 from adults, according to a Singapore study of household transmission of the virus.
The study by KK Women’s and Children’s Hospital (KKH) drew on data of children who were household contacts of COVID-19 cases and were screened for the disease at the hospital between March and April. A total of 213 children under the age of 16 were screened for COVID-19 during this period. They came from 137 households with a total of 223 adults who were confirmed cases, according to the study. Of the 213 children evaluated, 13 cases of adult-to-child COVID-19 transmission were detected across seven households.
This amounted to an “attack rate” of 6.1 per cent among children and 5.2 per cent of the households with confirmed exposure to COVID-19, the study found.
Further analysis showed that the adult-to-child transmission rate for children under the age of five was 1.3 per cent. This was the lowest rate among all age groups, compared to 8.1 per cent for children aged five to nine, and 9.8 per cent for children aged 10 to 16.
The findings were published in October in the Journal of Pediatrics.
The study also found that the risk of secondary infection in children was highest if the index COVID-19 patient was the child’s mother. At 11.1 per cent, the rate was almost double that if the index case was a father or grandparent.
“Because population susceptibility to SARS-CoV-2 is assumed to be universal, the attack rate in children would be expected to be similar to that in adults,” the authors of the study wrote.
“Because transmission is known to be correlated with degree of contact, attack rates might be expected to be higher in younger children, who presumably have closer interactions with their parents than do older children.
“However, in our study the attack rate was lowest in the youngest age group,” the study's authors wrote. Dr Yung Chee Fu, a consultant at KKH’s Infectious Disease Service and a co-author of the study, said that younger children might have more resistance to COVID-19 infection.
“It is possible that younger children are more resistant to SARS-CoV-2 infection at a cellular level,” he said in an Oct 13 newsletter about the study on KKH’s website. Dr Yung made reference to studies that have found a trend of “increased expression”, as age increases, of an enzyme receptor in the nasal epithelium that is used by the coronavirus for host entry.
The study also noted reports of a multisystem inflammatory syndrome in children infected with COVID-19, which has led to severe illness and death in some cases.
“The subpopulations of children at risk and the full spectrum of multisystem inflammatory syndrome in children remain unknown,” the study’s authors wrote. "However, the low attack rate among children in the youngest age group suggested that they “are less likely to become infected than adults and may not be drivers of the epidemic," they added.
“The low attack rate suggests that strict compliance with infection control may be able to eliminate or reduce the risk of transmission from adults to children in household settings."
ABOUT 4 IN 10 CHILDREN ASYMPTOMATIC
Another recent study by KKH of 39 children with COVID-19 in Singapore found that 38.5 per cent of the children remained asymptomatic.
This high proportion of asymptomatic cases made it difficult to identify children with COVID-19 based on symptomatic testing alone, said author Dr Li Jiahui.
“This important finding underscores the importance of early screening and isolation of children upon detection of potential exposure to an index case of COVID-19,” added Dr Yung, also a co-author of the study.
The study was published in August in the Annals, the official medical journal of the Academy of Medicine, Singapore.
It drew on data from 39 cases of children with COVID-19 who were admitted to KKH between January and May. This represented about 70 per cent of all pediatric cases detected in Singapore.
A low-grade fever, runny nose, sore throat and cough were the most common symptoms reported by children with COVID-19, according to the study. Other symptoms included diarrhoea and loss of smell or taste.
“Symptomatic children were more likely to have abnormal laboratory parameters but they did not have a poorer outcome compared to asymptomatic cases,” the study found.
All the children had a “mild course of illness” and were discharged “well”, with a mean hospital stay of 15 days, according to the study.
20 Oct 2020
COVID-19: SINGAPORE TO PILOT PRE-EVENT RAPID TESTING FROM MID-OCTOBER SO MORE EVENTS CAN SAFELY RESUME
SINGAPORE: Singapore will pilot pre-event testing from mid-October to December to identify a model that can be widely implemented so that more large-scale events can safely resume, announced Minister for Health Gan Kim Yong on Tuesday (Oct 20).
This means that participants going to an event or activity will be tested either at the event venue or at a separate testing facility, and only participants who have tested negative will be allowed to participate in the event.
These events could include business-to-business events and wedding receptions, added Mr Gan. For example, pre-event testing will be piloted at Singapore International Energy Week (SIEW) next week.
Antigen rapid tests (ARTs) will be used for the pre-event testing, which can return “fairly accurate” results within about half an hour, said the Ministry of Health (MOH) in a factsheet.
Although polymerase chain reaction (PCR) tests are currently used as the “definitive test” for confirming COVID-19 cases, the results take about one to two days, which is not feasible for pre-event testing, the ministry noted.
The ART results will be valid for 24 hours from the time of the swab, and it must be valid at the point of entry until the end of the event, said MOH in a technical briefing on Tuesday.
“If you go for a multi-day event, you would then require daily testing, with that cert being valid for 24 hours. However, if you’re attending multiple events within a 24-hour window, that same cert will gain you access to multiple events if you’re negative.”
Individuals who have been infected with COVID-19 and have recovered will be exempted from the pre-event testing within “a recent enough window” that ensures they are likely to be immune or protected from infection - for example, 180 days from the first positive PCR test, said MOH at the technical briefing.
Individuals who test positive on the ART at pre-entry testing must self-isolate and go for a PCR confirmatory swab, receiving referral forms that can be used to redeem the swab free of charge at clinics and regional screening centres, said the ministry. They also must not leave their place of isolation until they receive a negative PCR result, except to go for the test or seek medical attention.
For the ARTs that are being piloted, a nasal swab will be carried out to obtain a sample from the lower part of the nose and this should not be uncomfortable, said MOH. As part of the pilot, the ARTs and the PCR tests, where required, will be provided free of charge to the participants.
Depending on the event, participants will be informed by the event organiser on the specific requirements of the pre-event testing, as different processes will be tested for each pilot.
“For example, if the testing is being conducted at the event venue prior to admission, participants will likely have to arrive at the venue earlier to cater time for testing before attending the event,” said MOH in the factsheet.
“If the testing is conducted at a separate testing facility, participants will have to visit a separate testing facility beforehand and produce a valid certificate showing that the participant had tested negative for COVID-19 within a 24-hour timeframe from the end of the event, before being allowed to enter the event.”
20 OCT 2020
ROADMAP TO PHASE 3 ( MESSAGE FROM MOH )
It has been more than half a year since we entered Circuit Breaker, and more than four months since we started our gradual reopening. We have come a long way, ramping up healthcare capacity, contact tracing and testing capabilities, putting in place safe management measures in the community, workplaces and dormitories, and opening our borders in a safe and calibrated manner. With everyone’s efforts and sacrifices, we have been able to control the infection and keep community transmission low thus far.
To continue our journey of safe re-opening towards Phase Three, several conditions and enablers need to be in place. In particular, we must continue scaling up testing and tracing efforts as well as adhering to the safe management measures. This is how we can progressively resume more activities safely while keeping community transmission low.
Phase Three is not a return to the pre-COVID status quo ante. It will entail new ways of working and living, until the world has the virus under tight control (e.g. through more effective treatments and vaccines that are widely available). So we have to be prepared to stay in Phase Three for a prolonged period (potentially more than a year). Phase Three will also not be static. If we can put in place more enablers, there is scope for further reopening and scaling up of activities even within Phase Three.
Key Enablers for Phase Three
Continued Adherence to Safe Management Measures
We have seen a resurgence of COVID-19 in other countries as they open up to more activities or due to lack of adherence to safe management measures. To ensure that the situation in Singapore remains under control despite the continued presence of cases in our community, we will each have to keep up the habits and practices that we have painstakingly put in place over the past few months:
Testing to Enable More Activities
Small group sizes. Keeping group sizes small and limiting the number of contacts that we have slows down the rate of virus transmission and reduces the risk of super-spreading events. We will continue to set limits on group sizes, both in public spaces and visitors to the home.
Safe distancing. Safe distancing, including no intermingling between individuals and groups, remains a key measure in reducing the risk of spread. We will continue to set requirements on safe distancing, as well as to disallow or minimise interactions between groups.
Social responsibility. Mask-wearing and personal hygiene protect us and others from COVID-19. Persons with symptoms of acute respiratory infection should seek medical attention at the earliest opportunity and follow up with a Polymerase Chain Reaction (PCR) swab test if directed by the doctor.
Testing to Enable More Activities
To enable more activities to resume in a safe manner, the Multi-Ministry Taskforce is piloting the use of pre-event COVID-19 testing for larger-scale and higher-risk activities to reduce the risk of a COVID-19 case being present at the event.
Pre-event testing requires participants to an event or activity to be tested shortly before the event, either at the event venue itself or at a separate testing facility. Only participants who test negative for COVID-19 will be allowed to participate in the event. This helps to prevent positive cases from transmitting the disease to other people at the event.
Given the need for a shorter turnaround time for pre-event testing, we are exploring the use of antigen rapid tests which can return fairly accurate results quickly. This is part of our strategy to expand beyond the highly accurate PCR tests to have a range of tests to cater for various use cases. Due to the lower sensitivity of antigen rapid tests, there is still a possibility that a COVID-19 case could slip through to attend the event. Therefore there is still a need for safe management measures to be put in place, including mask-wearing, safe distancing, group size and capacity limits, to reduce the risk of transmission. The lower accuracy of antigen rapid tests also means there could be false positives, i.e. persons who are tested positive even though they are not infected.
The pilots will enable the Ministry of Health (MOH) to trial pre-event testing processes to identify different models that can be implemented more widely and allow more large-scale or higher-risk events to resume eventually. From mid-October to December 2020, we will be identifying selected events across different settings, such as business-to-business events, wedding receptions, live performances, and sports events to test different operational workflows for pre-event testing. For example, one business-to-business event where pre-event testing will be piloted is the Singapore International Energy Week next week. We will gather feedback from event organisers and participants to finetune these processes. If these pilots prove successful, we will assess how to make these tests available for more widespread use. For further information, please refer to this Factsheet on Pilots for Pre-event Testing.
Digital Contact Tracing Tools to Enable More Activities
Alongside safe management measures and scaled up testing, we need to continue to strengthen our contact tracing regime, leveraging our two key digital contact tracing tools – SafeEntry and TraceTogether. The active use of SafeEntry has significantly improved MOH’s ability to detect COVID-19 clusters quickly. At the same time, we need greater participation in the TraceTogether Programme to help us quickly identify close contacts of COVID-positive cases. These digital tools are critical for quickly ringfencing any cases that emerge as a result of larger group sizes and larger-scale activities. Without these tools, we may have multiple generations of spread and large clusters forming.
We will need a higher take-up rate for TraceTogether before we can start Phase Three. Local and international experience suggests that social interactions, which often take place in close proximity, sometimes without masks on, and often for prolonged periods, are key areas of spread. It is therefore imperative that there is higher adoption of TraceTogether, so that we are able to quickly contact trace and ringfence any infections.
Another prerequisite is to expand the deployment of TraceTogether-only SafeEntry, where visitors are required to check in to SafeEntry through either scanning the QR code using the TraceTogether App on your phone, or by presenting your TraceTogether Token to be scanned. This allows us to ensure that persons visiting places where people are likely to be in contact for prolonged periods, or where human traffic is high, have the TraceTogether App or Token. In this regard, we will progressively roll out TraceTogether-only SafeEntry to workplaces, schools, and more public venues, including cinemas, live performances, shopping malls, and F&B outlets.
We encourage everyone to download the App or collect a Token for use at the TraceTogether-only SafeEntry premises. TraceTogether tokens are now available for collection at 38 Community Centres/ Clubs (CCs) around Singapore, and are expected to be available at all 108 CCs by end-November 2020. Please refer to the TokenGoWhere website (https://token.gowhere.gov.sg) to find the latest schedule and locate a convenient site for collection.
Moving to Phase Three
With the above conditions and enablers in place, we will be able to move to Phase Three. The shift to Phase Three will entail the following adjustments. The exact timing of these changes will depend on our collective ability to cooperate with the requirements. Details of some of the measures which will be updated in the coming weeks are summarised in Annex.
Increasing Group Size
In Phase Three, the group size for gatherings outside the home could be increased from the present 5 persons to 8 persons. The number of visitors allowed to homes would similarly increase to 8.
Increasing Capacity Limits
Presently, public venues like museums and attractions are already open, subject to a capacity limit. Events like congregational worship services and wedding receptions can also be held in two zones of 50 persons. In Phase Three, these capacity limits could be increased, and we could allow events with multiple zones of 50 persons. All of these adjustments would have to be done in a controlled manner, setting by setting, over the course of Phase Three, and additional measures would apply for specific settings.
For example, wedding receptions are higher-risk activities, where people are gathered for a meal without their masks on, and there is a greater likelihood of social interactions. Any expansion in the number of attendees would therefore require additional safety measures, including having all the guests go through a pre-event test.
It is also critical to adhere to the safe management measures in these large-scale events. Therefore, Government agencies may also request video and photographic footage of the events, to facilitate checks and investigations against any breaches of the rules.
Considerations for Activities in Higher-Risk Settings
Higher-risk settings like bars, pubs, karaoke lounges and nightclubs are closed today as their activities pose a higher risk of transmission. Even at the start of Phase Three, we do not expect to open these venues. But we are prepared to consider a few limited pilots in these settings, subject to a more stringent set of measures, including pre-entry testing, to explore how the industry could resume safely. We will discuss these possibilities with the nightlife industry. But we have to be realistic that it will take a long time for the industry to fully re-open in its original form. Where there is non-resumption, the Government will put in place an assistance package to help business operators and owners transit and pivot to new areas.
Facilitating travel and re-opening of borders
Unlike larger countries which can keep their borders closed, our livelihoods and economic survival depend heavily on Singapore being open to the world and being a key international and travel hub. It is therefore not possible for Singapore to keep our borders closed indefinitely. We will have to gradually allow more travel to resume in a safe manner.
Through the unilateral opening of borders and Air Travel Bubbles, we are allowing travellers from low-risk countries/ regions (i.e. those which have comprehensive public health surveillance systems and displayed successful control over the spread of the COVID-19 virus) to enter Singapore with a COVID-19 test without the need to serve a Stay-Home Notice (SHN).
For travellers from higher-risk countries/ regions, our current approach is to subject them to SHN at home or at dedicated SHN facilities. We are exploring ways to deploy more frequent testing, coupled with other safeguards, to enable more travellers to enter Singapore without a need to serve SHN, while minimising the risk of transmission to the community.
Currently, Singaporeans Citizens (SC)/ Permanent Residents (PR)/ Long-Term Pass Holders (LTPH) who travel out of Singapore from 27 March 2020 onwards are responsible for their own inpatient medical bills, if they have onset of symptoms for COVID-19 within 14 days of their return to Singapore, and are unable to access government subsidies or insurance coverage (be it MediShield Life/ Integrated Plans or private insurance). [1] In line with the progressive move to reopen our borders, we will henceforth allow all SCs/ PRs/ LTPHs travelling overseas to access government subsidies and insurance coverage for their medical bills.
There is no change to the charging policy for SCs/ PRs/ LTPHs who departed Singapore before 27 March 2020, i.e. the Government will continue to pay for their inpatient medical bills for COVID-19 treatment upon return, if any.
Road to Phase Three and Beyond
As we make these adjustments towards a new COVID normal, the Multi-Ministry Taskforce will continue to explore parallel tracks such as vaccines, treatment, testing and other technologies which could enable us to further ease restrictions and resume connectivity with the world in a safe manner. The journey ahead may be long, but with the cooperation of everyone, including businesses, scientists, frontline workers, and the general public, we are confident of transiting smoothly to Phase Three and beyond, emerging stronger from this experience.
19 Oct 2020
ENABLE FURTHER SAFER REOPENING BY COMBINING THE STRENGTHS OF SAFEENTRY (SE) AND THE TRACETOGETHER (TT) PROGRAMME VIA TT-ONLY SE
Singapore, 20 October 2020 – In order to resume larger-scale activities and further reopen our economy in a safer manner, TT-only SE will be progressively expanded to more venues. The use of TT-only SE will provide added assurance that everyone present at these large scale activities are better protected by effective contact tracing through participation in the TT Programme.
TT-only SE ensures that if a COVID-19 case is identified, we can quickly inform close contacts in those locations through the TT Programme. The close contacts can immediately take the necessary precautions to keep their loved ones safe.
By end of this year, we plan for all popular venues that currently require SE to transit to TT-only SE. The current modalities of i) scanning on-site QR codes with a phone camera, ii) using SingPass Mobile for SE check-in, and iii) scanning of personal IDs, will be disabled at venues where TT-only SE is implemented. Instead, SE is allowed only if one uses their TT App to scan the on-site QR code, or if they let the on-site entry staff scan the QR code on their TT Token.
COVID-19 OVERSEAS NEWS
25 Oct 2020
US SEES RECORD COVID-19 NUMBERS FOR SECOND STRAIGHT DAY
WASHINGTON: The United States saw a record high number of new daily COVID-19 cases for the second day in a row on Saturday (Oct 24), figures from Johns Hopkins University showed, as warnings grew over its spread.
The country reported 88,973 new infections between 8.30pm Friday and 8.30pm Saturday local time, the figures showed, substantially above the previous day's 79,963.
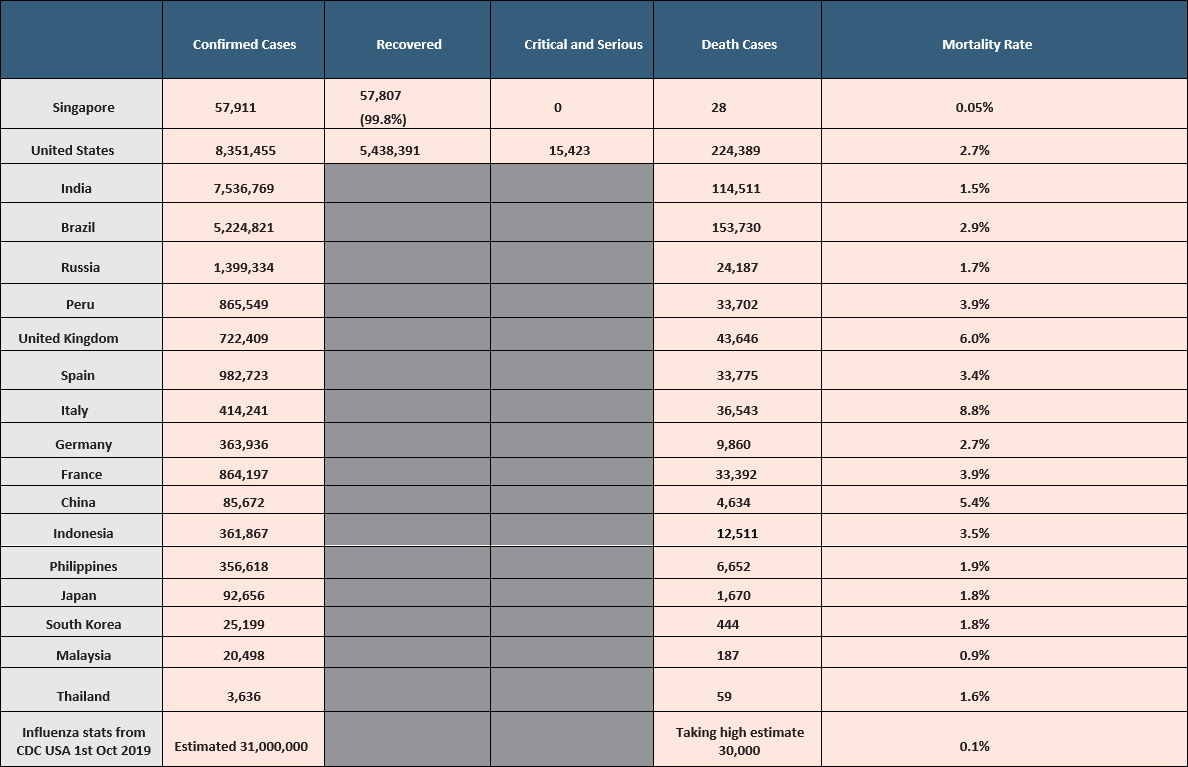
A total of 8,568,625 cases have been reported in the United States with 224,751 deaths, the highest in the world in absolute terms.
The worst current outbreaks in the country are in the north and Midwest, and some 35 of the 50 states are seeing an increase in case numbers.
The number of deaths over 24 hours has remained broadly stable since the beginning of autumn, with between 700 and 800. On Saturday, the US recorded 906 deaths, the Johns
25 Oct 2020
CHINA REPORTS 15 NEW COVID-19 CASES, ALL IMPORTED INFECTIONS
BEIJING: China reported 15 new COVID-19 cases in the mainland for Oct 23, down from 28 cases in the previous day, the health commission said on Sunday (Oct 25). All 15 of the new infections were imported, according to a statement by the National Health Commission. It also reported 19 new asymptomatic patients, compared with 27 a day earlier. As of Saturday, mainland China had 85,790 confirmed coronavirus cases, the health authority said. The COVID-19 death toll stands at 4,634.
24 Oct 2020
MALAYSIA REPORTS 1,228 NEW COVID-19 CASES, BIGGEST DAILY JUMP
KUALA LUMPUR: Malaysia reported 1,228 new coronavirus cases on Saturday (Oct 24), a record daily jump since the start of the pandemic, bringing the total number of cases to 25,742.
The state of Sabah had the highest number of cases on Saturday - 449 infections. The remaining affected states are: Selangor, Pulau Pinang, Perak, Negeri Sembilan, Sarawak, Kedah, Melaka, Terengganu and Pahang.
The health ministry also said seven new deaths were recorded across the Southeast Asian country. This takes Malaysia's national death toll to 221 cases.
A total of 92 COVID-19 patients are still in hospital, with 31 of them on ventilators.
Saturday's figures came amid speculation of a possible state of emergency in Malaysia - a measure opposition politicians have cried foul over.
24 Oct 2020
ASIA BECOMES SECOND REGION TO EXCEED 10 MILLION COVID-19 CASES
REUTERS: Asia surpassed 10 million infections of the new coronavirus on Saturday (Oct 24), the second-heaviest regional toll in the world, according to a Reuters tally, as cases continue to mount in India despite a slowdown and sharp declines elsewhere.
Behind only Latin America, Asia accounts for about one-fourth of the global caseload of 42.1 million of the virus. With over 163,000 deaths, the region accounts for some 14 per cent of the global COVID-19 toll.
The Reuters tally is based on official reporting by countries. The true numbers of cases and deaths are likely much higher, experts say, given deficiencies in testing and potential under-reporting in many countries.
Despite the Asian spikes, the region overall has reported improvement in handling the pandemic in recent weeks, with daily caseloads slowing in places like India - a sharp contrast to the COVID-19 resurgence seen in Europe and North America.
Within the region, South Asia led by India is the worst affected, with nearly 21 per cent of the reported global coronavirus cases and 12 per cent of deaths. This contrasts with countries like China and New Zealand that have crushed infections and Japan, where COVID-19 had been stubbornly entrenched but not accelerating.
India is the worst-hit country in the world after the United States, although infections are slowing in the world's second-most populous country. India is reporting more than 57,000 cases of the virus a day, viewed on a weekly average, with 58 new cases per 10,000 people in Asia's third-largest economy, according to a Reuters analysis.
India is averaging 764 COVID-19 deaths a day, the worst in the world and accounting for one in every 13 global pandemic deaths.
The country has reported nearly 7.8 million infections, behind the US tally of 8.5 million, and nearly 118,000 deaths, versus 224,128 in the United States. Unlike the recent US surge, however, India's slowdown saw the lowest daily caseload in nearly three months on Wednesday.
However, India's infections may surge again, doctor’s fear, with a holiday approaching and winter bringing more severe pollution from farmers burning stubble, worsening the breathing difficulties that many COVID-19 patients suffer.
India's eastern neighbour Bangladesh is Asia's second-worst hit country, with nearly 400,000 cases. But daily infections have slowed to 1,453, less than 40 per cent of the July peak.
In Southeast Asia, Indonesia surpassed the Philippines last week as the worst-hit nation with more than 370,000 infections.
The world's largest Muslim-majority nation, Indonesia has struggled to get its outbreak under control.
The Philippines, which last week reported its biggest daily tally in a month, has put partial coronavirus restrictions around the capital Manila until Oct 31 to check COVID-19.
Despite Asia's patchy record, a World Health Organization expert said on Monday that Europe and North America should follow the example of Asian states in persevering with anti-COVID measures and quarantine restrictions for infected people.
Mike Ryan, head of the UN agency's emergencies programme, said the global death toll from COVID-19 could double to 2 million before a successful vaccine is widely used and could be even higher without concerted action to curb the pandemic.
24 Oct 2020
SOUTH KOREA REPORTS 77 COVID-19 CASES TIED TO HOSPITALS
SEOUL: South Korea has reported 77 new cases of the coronavirus, mostly from the greater capital area where officials are scrambling to stem transmissions at hospitals and nursing homes. Figures released by the Korea Disease Control and Prevention Agency on Saturday (Oct 24) brought the country’s caseload to 25,775, including 457 deaths. Among the 1,484 active cases, 60 are in serious condition.
Fifty-nine of the new cases were reported from densely populated Seoul metropolitan area, which has emerged as the epicentre of the outbreak since summer.
Hundreds of cases have been linked to a handful of hospitals and nursing homes. Officials are testing thousands of medical workers to stem infections. Eleven of the new cases were tied to international arrivals, including passengers from the United States, the Philippines and India.
24 Oct 2020
ASTRAZENECA, JOHNSON & JOHNSON COVID-19 VACCINE TRIALS BACK ON TRACK IN US
WASHINGTON: Two major clinical trials for experimental COVID-19 vaccines got back on track in the United States on Friday (Oct 23), providing a glimmer of hope as the number of cases skyrockets across the country.
AstraZeneca announced that the trial of its vaccine candidate, developed with Britain's University of Oxford, has resumed in the US, the only country where it remained suspended following a participant's illness six weeks ago.
"The Food and Drug Administration (FDA) today authorised the restart in the US, following the resumption of trials in other countries in recent weeks," the drug maker said.
The trial was suspended worldwide on September 6, but resumed shortly thereafter in Britain, and in the following weeks in South Africa, Brazil and Japan, with authorities determining the illness was not apparently linked to the vaccine.
"The FDA reviewed all safety data from trials globally and concluded it was safe to resume the trial," AstraZeneca said.
The company added it was hoping to have results later this year, "depending on the rate of infection within the communities where the clinical trials are being conducted".
The AstraZeneca vaccine project is one of the most promising and advanced in the world to combat the global pandemic, which has now claimed the lives of 1.1 million people. It is one of 10 vaccine candidates being tested on tens of thousands of people in so-called phase 3 trials.
In the United States, the two top candidates vying to get a green light from the FDA are those made by Pfizer and Moderna. Both expect to request approval next month. Many countries are counting on using the AstraZeneca vaccine to inoculate their populations. The drug maker pre-sold hundreds of millions of doses on several continents and signed partnership deals with other producers to ensure the doses could be made locally.
"NO EVIDENCE" VACCINE CAUSED ILLNESS
Shortly after AstraZeneca's announcement, Johnson & Johnson said it was preparing to resume recruitment for its parallel trial, which was suspended last week after a volunteer fell ill.
"After a thorough evaluation of a serious medical event experienced by one study participant, no clear cause has been identified," the group said in a statement. "The company has found no evidence that the vaccine candidate caused the event."
24 Oct 2020
WHO'S TEDROS SAYS COUNTRIES ON 'DANGEROUS TRACK' IN COVID-19 PANDEMIC
GENEVA: The world is now at a critical juncture in the COVID-19 pandemic and some countries are on a dangerous path, facing the prospect of health services collapsing under the strain, the head of the World Health Organization said on Friday (Oct 23).
"We are at a critical juncture in the COVID-19 pandemic, particularly in the Northern hemisphere," WHO Director-General Tedros Adhanom Ghebreyesus told a news conference.
"The next few months are going to be very tough and some countries are on a dangerous track."
"We urge leaders to take immediate action, to prevent further unnecessary deaths, essential health services from collapsing and schools shutting again. As I said it in February and I’m repeating it today: This is not a drill."
Tedros said too many countries were now seeing an exponential increase in infections, "and that is now leading to hospitals and intensive care units running close or above capacity - and we’re still only in October".
He said countries should take action to limit the spread of the virus quickly. Improving testing, tracing of contacts of those infected and isolation of those at risk of spreading the virus would enable countries to avoid mandatory lockdowns.
Several countries in Europe are reporting infection rates higher than during the first wave of the pandemic in March and April, with Spain saying it has now more than three million cases.
Governments across the continent are slapping urgent new restrictions on daily life, with France extending a curfew to cover 46 million people and Ireland locked down again.
"The continuing increases in COVID-19 infections ... pose a major threat to public health, with most countries having a highly concerning epidemiological situation," said Andrea Ammon, director of the European Centre for Disease Prevention and Control (ECDC).
The agency said all EU countries except Cyprus, Estonia, Finland and Greece fell into a "serious concern" category, as did the United Kingdom, up from just seven a month ago.
Across the planet, COVID-19 has now claimed the lives of 1.1 million people - about one fifth in the United States - and infected close to 42 million.
22 Oct 2020
US COVID-19 HOSPITALISATIONS HIT TWO-MONTH PEAK
WISCONSIN: The number of COVID-19 patients in US hospitals hit 40,000 for the first time since August on Wednesday (Oct 21), according to a Reuters tally, as the nation battles a surge in infections led by Midwest states.
Hospitals have seen a 36 per cent rise in coronavirus patients over the past four weeks and Midwest hospitals are setting new records every day.
So far in October, 16 states have reported their highest daily numbers of hospitalised COVID-19 since the pandemic started, including the Midwest states of Iowa, Kansas, Missouri, Nebraska, North Dakota, South Dakota and Wisconsin.
Hospitalisations of virus-stricken patients have set records in every region except the Northeast. Hospitalisations are a closely watched metric because they are not influenced by how much testing is done.
In addition to hospitalisations reaching 40,264 on Wednesday, the seven-day average of new cases of COVID-19 have risen by 45 per cent in the past four weeks and is approaching levels last seen during the summer peak, according to a Reuters analysis.
On Friday, the US recorded 69,478 new cases, the highest single-day total since July 24 and the fifth-highest single-day total since the start of the pandemic.
22 Oct 2020
US LIKELY TO HAVE ENOUGH COVID-19 VACCINES FOR ALL VULNERABLE AMERICANS BY YEAR END: OFFICIAL
WASHINGTON: The United States is likely to have enough safe and effective COVID-19 vaccines available to inoculate the most vulnerable Americans by the end of 2020, Health and Human Services Secretary Alex Azar said on Wednesday (Oct 21).
The US government is "cautiously optimistic" that one or two vaccines, likely from Pfizer or Moderna, will be available by the end of the year and can begin to be distributed to Americans, officials said during a news conference. Azar said he expects all seniors, healthcare workers, and first responders will be able to receive a vaccine as soon as January, with the rest of the American public able to get a vaccine by April.
Companies participating in the US government's effort to develop a vaccine for COVID-19, dubbed Operation Warp Speed, have begun developing manufacturing capabilities even before any vaccinations have been authorised by regulators. In an open letter published last week, Pfizer said it is unlikely to have enough data to submit for a US regulatory authorisation until late November, after the US presidential election.
UPDATES ON LATEST CONFIRMED CASES IN SG
As of 25 October 2020, 12pm, the Ministry of Health (MOH) has confirmed and verified 2 new cases of locally transmitted COVID-19 infection. Both reside in dormitories and there are no new cases in the community. In addition, there are 3 imported cases, who had all been placed on Stay-Home Notice (SHN) upon arrival in Singapore. Amongst the new cases today, 4 are asymptomatic, and were detected from our proactive screening and surveillance, while 1 was symptomatic.
| Number of cases | Already in quarantine/ isolation before detection | Detected from surveillance | Symptomatic | Asymptomatic | |
| Cases in the community | 0 | 0 | 0 | 0 | 0 |
| Cases residing in dormitories | 2 | 0 | 2 | 1 | 1 |
| Imported cases | 3 | 3 | 0 | 0 | 3 |
| Total | 5 | 3 | 2 | 1 | 4 |
Overall, the number of new cases in the community has decreased from 5 cases in the week before, to 2 in the past week. The number of unlinked cases in the community has remained stable at 2 cases a week in the past 2 weeks.
Case Details
Cases in the community: 0
There are no cases in the community today.
Cases residing in dormitories: 2
There are 2 cases residing in dormitories today, of whom 1 was detected from the bi-weekly Rostered Routine Testing of workers living in dormitories, even though he is asymptomatic. The other was tested for COVID-19 when he developed acute respiratory infection (ARI) symptoms.
Imported cases: 3
Amongst the 3 imported cases, 1 (Case 58140) is a Singapore Permanent Resident who returned from the UK, and another (Case 58142) is a Dependant’s Pass holder who arrived from the UAE. The remaining case (Case 58143) is a Short-Term Visit Pass holder who arrived from Indonesia. She was allowed entry into Singapore to visit her child who is a Singapore Permanent Resident. They were placed on Stay Home Notice (SHN) upon arrival in Singapore and were tested while serving their SHN.
Epidemiological investigations are in progress. In the meantime, all the identified close contacts of the cases have been isolated and placed on quarantine, and will be tested at the start and end of their quarantine period. Serological tests for the close contacts will be conducted to determine if the cases could have been infected by them.
MOH has been monitoring existing clusters for any further transmission. As there have been no more cases linked to the cluster at North Coast Lodge (51 North Coast Avenue) for the past two incubation periods (i.e. 28 days), the cluster has now been closed.
Update on condition of confirmed cases
14 more cases of COVID-19 infection have been discharged from hospitals or community isolation facilities. In all, 57,858 have fully recovered from the infection and have been discharged from hospitals or community care facilities.
There are currently 51 confirmed cases who are still in hospital. Of these, most are stable or improving, and none is in the intensive care unit. 33 are isolated and cared for at community facilities. These are those who have mild symptoms, or are clinically well but still test positive for COVID-19. 28 have passed away from complications due to COVID-19 infection.
Summary of Current Cases in SG
| CONFIRMED CASES IN SINGAPORE ( as of 25 Oct 2020 ) | 57, 970 |
| HOSPITALISED | 51 |
| RECOVERING IN COMMUNITY FACILITIES | 33 |
| CASES IN CRITICAL CONDITION | 0 |
| DISCHARGED | 57,858 |
| DEATH | 28 |
Stats adapted from Worldometer as of 0100 26 Oct 2020